Sutures Needles
Category
NEED MORE
INFORMATION
CONTACT US
Biotronix Healthcare Customer Service Associates are available 8:30am to 5:pm EST Monday thru Friday
North America Customer Service
Tel: (954) 320-6088
(954) 440-1572
customerservice@biotronixhealthcare.com
International Customer Service
Tel: (289) 813-0159
(416) 876-5035
customerservice@biotronixhealthcare.com
______________________________________
Sales and Orders
Tel: (954) 266-8944
sales@biotronixhealthcare.com
______________________________________
Regulatory Affairs
regulatory@biotronixhealthcare.com
______________________________________
Accounting Department
accounting@biotronixhealthcare.com
______________________________________
Call to speak with a knowledgeable Biotronix Healthcare specialist today.

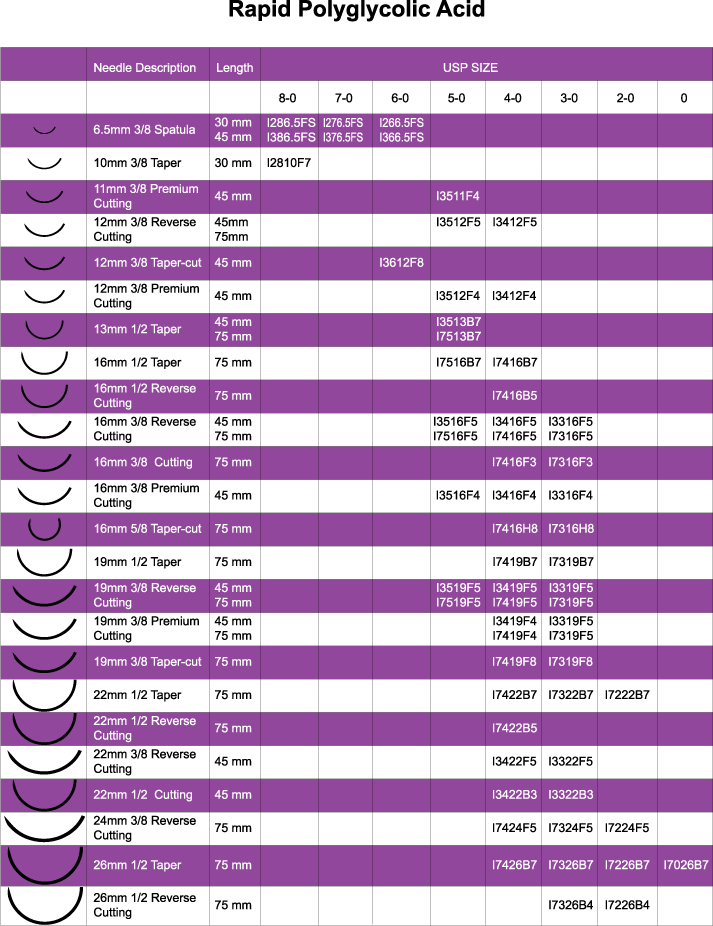
Rapid Polyglycolic Acid
Biotronix Healthcare Polyglycolic Acid sutures are synthetic absorbable sutures composed of polyglycolic acid and a calcium stearate and polycaprolactone coating.
Biotronix Healthcare Polyglycolic Acid sutures has minimal tissue reactivity during the absorption process, superior tensile strength and excellent knotting security. The braided construction ensures that handling and knotting are excellent and the unique lubricant coating remains effectively bonded to the material throughout its use, ensuring smooth passage through tissue and easy knot tie down.
Biotronix Polyglycolic Acid sutures hold more than 75% of its tensile strength at two weeks and complete absorption takes place in approximately 60 to 90 days.

Applications:
Indicated for use in soft tissue approximation where only short-term wound support is required and where the rapid absorption of the suture would be beneficial. Due to its absorption profile Rapid Polyglycolic acid (PGA) is useful for skin closure, particularly in pediatric surgery, episiotomies, circumcisions and closure of oral mucosa.
Characteristics of Biotronix Healthcare Rapid Polyglycolic Acid sutures:
• Retention of 50% knot tensile strength after 7 days.
• Complete mass absorption in around 42 days.
• High initial tensile strength
• Predictable and constant degradation rate
• Good knot security
• Excellent handling properties
• Quick mass absorption
Packaging:
Individually packaged 12 or 36 per box. Also available in reels.
Contraindications:
Due to the rapid loss of tensile strength, the Rapid Absorbable Surgical Suture should not be used where extended approximation of tissues under stress is required or where wound support beyond 7 days is required. The Rapid Absorbable Surgical Suture is not for use in ligation, cardiovascular and neurological tissues.
Warnings / Precautions:
Do not use if package is open or damaged or if the expiration date has been exceeded. Discard open, unused suture. Do not re-sterilize. Re-sterilization may alter the physical properties Users should exercise caution when handling surgical needles to avoid inadvertent needle sticks. Discard used needles in a “sharps” container.
Avoid storing product at elevated temperatures. As with any foreign body, prolonged contact of this or any other suture with salt solutions, such as those found in the urinary or biliary tracts, may result in calculus formation. Acceptable surgical practice should be followed with respect to drainage and closure of contaminated or infected wounds.
The use of supplemental non-absorbable sutures should be considered by the surgeon in the closure of sites which may undergo expansion, stretching, or distention, or which may require additional support as this is an absorbable suture material.
Adverse Reactions:
Adverse reactions associated with the use of this device include transitory local irritation at the wound site, transitory inflammatory foreign body response, erythematic and indurations during the absorption process of subcuticular sutures. Like all foreign bodies this suture may potentates an existing infection.